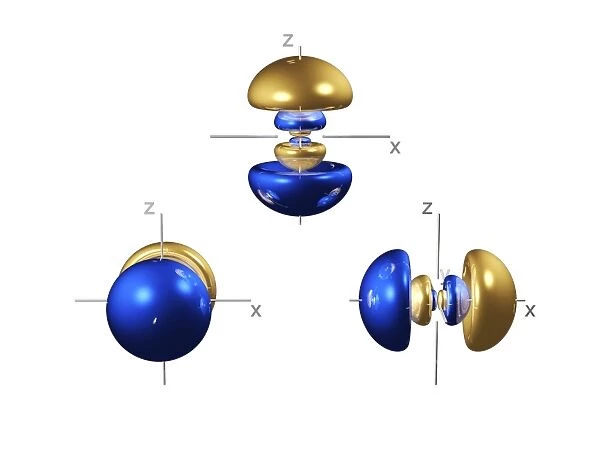
4p electron orbitals
![]()

Wall Art and Photo Gifts from Science Photo Library
4p electron orbitals
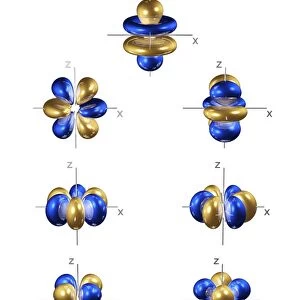
2p electron orbitals, computer model. An electron orbital is a region around an atomic nucleus (not seen) in which one or a pair of electrons is most likely to exist. The three 2p orbitals have a bi- lobed shape centred on the nucleus. They are part of the 2 shell, which also contains a spherical, lower energy 2s orbital. In atoms, electrons fill the lower energy orbitals first. The 2s orbital is only filled when the lowest energy 1s orbital is full, and the 2p orbitals only when both 1s and 2s are full. The 1s and 2s orbitals can both hold a pair of electrons each, as can each 2p orbital. The 2p orbitals are thus full in element 10, neon. Neons electron configuration is written 1s2 2s2 2p6
Science Photo Library features Science and Medical images including photos and illustrations
Media ID 6291249
© DR MARK J. WINTER/SCIENCE PHOTO LIBRARY
Atom Atomic Axes Axis Configuration Electron Electron Orbital Mechanical Orbital Orbitals Quantum Mechanics Quantum Physics Shell Shells Trio Computer Artwork Physical
EDITORS COMMENTS
This print from Science Photo Library showcases the intricate beauty of 4p electron orbitals, specifically 2p electron orbitals. In this computer model, we are presented with a visual representation of the region surrounding an atomic nucleus where one or a pair of electrons is most likely to exist. The three 2p orbitals depicted in this image possess a distinct bi-lobed shape that is symmetrically centered on the nucleus. These orbitals belong to the 2 shell, which also encompasses a lower energy spherical 2s orbital. It is important to note that electrons fill the lower energy orbitals before occupying higher energy ones. Consequently, the filling sequence dictates that only when both the lowest energy 1s and 2s orbitals are full will the 2p orbitals be filled. Each of these remarkable electron shells can accommodate pairs of electrons - two for both the 1s and 2s orbital, as well as each individual 2p orbital. As such, element number ten on our periodic table, neon (Ne), possesses completely filled and satisfied sets of these fascinatingly shaped p-orbitals. This awe-inspiring model serves as an illustrative tool for understanding quantum mechanics and quantum physics within atoms' complex configurations. The mechanical precision exhibited in this computer artwork allows us to delve into the realm of atomic structure while appreciating its inherent beauty and significance in chemistry's fundamental principles.
MADE IN THE USA
Safe Shipping with 30 Day Money Back Guarantee
FREE PERSONALISATION*
We are proud to offer a range of customisation features including Personalised Captions, Color Filters and Picture Zoom Tools
SECURE PAYMENTS
We happily accept a wide range of payment options so you can pay for the things you need in the way that is most convenient for you
* Options may vary by product and licensing agreement. Zoomed Pictures can be adjusted in the Cart.